Mass Spectroscopy
Mass Spectrometry
The mass spectrometer is useful to analyse solids liquids and gases, each of these phases have four features in common.
• The section in which the sample is introduced is operated with a high vacuum, maintained with a continuous pump. Pumping is necessary to prevent the loss of ions from the sample by the accidental collisions with molecules in the atmosphere.
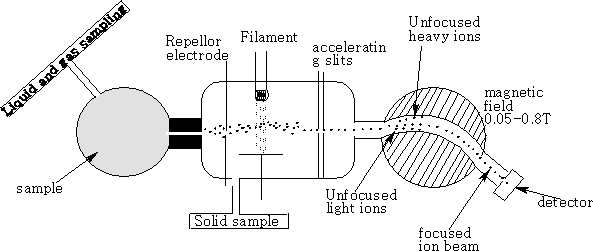
• An ion source:
Ions of the sample become ionised here. There are different methods by which this is performed
a) Bombardment with electrons from a filament; the electrons have an energy about 70eV. The energy absorbed by the molecules promotes ionisation by loss of electrons from the non-bonding or bonding orbitals, ions formed by the removal of one electron from the original molecule are called molecular or parent ions.
b) Desorption of ions from a surface by the action of an electric field.
c) Irradiation by UV light.
For solid materials:
d) Surface ionisation: ions are emitted from inorganic materials by evaporation when the material is heated to a high temperature
e) Spark source: Used for metals or other conductors in which a spark is produced between two electrodes made from the sample.
• An ion analyser or separator:
Beams of ions are produced from the ion source and separated into individual beams containing ions of a single mass. An accelerator plate with a positive potential of about 2000V is used to accelerate the positive ions down the tube.
This separation is achieved:
a) Passage through a magnetic field; ions are deflected in the magnetic field to different extents depending on their mass/charge ratio.
b) Passage through an electric field.
c) Measurement of the time it takes for different ions to move through an electric field.
• An ion collector: the beam of ions which passes through the separator is collected on an electrode where it picks up an electron to neutralise the positive charge, and the ion current which is produced is amplified and recorded as a function of the mass/charge ratio.